
If ∆S is negative, then the negative signs (from the subtraction and the sign of ∆S) will cancel out, and so as T∆S gets bigger, ∆G will get more positive. T is always positive, so if ∆S is positive then a bigger T∆S will make ∆G more negative (since we subtract T∆S). As T increases, the T∆S component gets bigger. Question: Predict the sign (positive, negative, or zero) of S S. 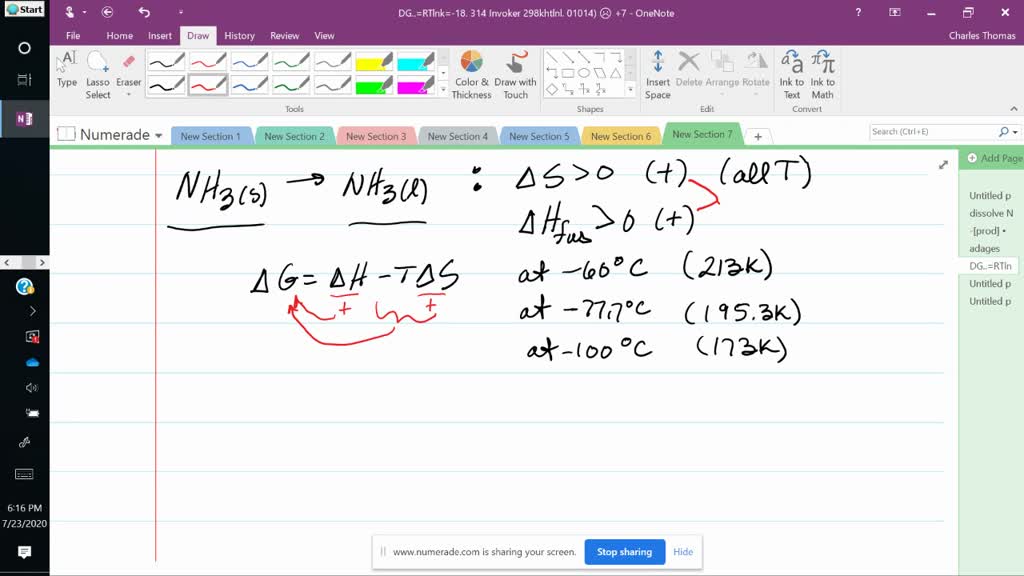
∆H is still positive and ∆S is still whatever sign you figured out above. Science Physics Entropy Predict the sign (positive, negative, or zero) of Delta S for the following reaction. Since ∆H and ∆S don't change significantly with temperature (given in the question), we can assume that they keep the same signs and values: i.e. C2H2(g) O2(g) arrow 2CO2(g) H2O(l) Delta H -1299.6 kJ C(s) O2(g) arrow CO2(g) Delta H -393.5 kJ H2(g) O2(g) arrow H2O(l) Delta H -285.8 kJ Calculate Delta Hrxn for the following reaction: CH4(g) 2Cl2(g) arrow CH2Cl2(g) 2HCl(g) Use the following reactions and given Delta Hs. If ∆G is negative (from the question), is the reaction spontaneous or non-spontaneous?Ģ) Let's use ∆G = ∆H - T∆S again.  
G < 0, the reaction is spontaneous in the forward direction, thermodynamically favourable. The change in enthalpy, Delta H, is equal to the sum of. For a given reaction if: G > 0, the reaction is nonspontaneous in the forward direction, not thermodynamically favourable. Course 32K views Enthalpy Change Formula Enthalpy, H, is a thermodynamic property that describes the heat lost or gained in a system. From these values, we can know for certain whether ∆S is positive or negative (hint: remember that we are subtracting ∆G!).ġ) Knowing the sign of ∆G is enough to say whether the reaction is spontaneous or not under these conditions. Look at it from the point of view of Gibbs Free Energy, G H T S. So for this question Im thinking I use the equation G H T S Then plug in the values and solve for T 8.9 k J 58.1 k J T ( 0. Calculate the temperature at which the reaction becomes spontaneous. The chemical equation is: 2 NO 2 (g) -> N 2 O 4 (g) For this reaction, E is -54.72 kJ. It is the entropy term that favors the reaction. Temperature is always positive (in Kelvin). 1 / 32 Predict the signs of delta S and delta H for the freezing of water into ice at 273 degrees Click the card to flip Definition 1 / 32 E. At 25 C, 298 K, the reduction of copper (I) oxide, H 58.1 k J, S 165 J / K, is nonspontaneous, G 8.9 k J. Consider first an endothermic reaction (positive (Delta H)) that also displays an increase in entropy (positive (Delta S)). Copy IUPAC Standard InChIKey: MYMOFIZGZYHOMD-UHFFFAOYSA-N Copy CAS. Chemistry Chemistry in society Revise Test 1 2 3 4 5 6 7 8 Calculating enthalpy changes The enthalpy change for a reaction can be calculated using the following equation: \ \Delta. Predict if the reaction would be spontaneous at 298 K and if not, at what temperature the reaction would become. Science Chemistry Chemistry questions and answers For the reaction of SO2 with O2 to form SO3 over the range of temps 300-1000K, calculate the delta H and delta S This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts. We know (from the question) that ∆G is negative and that ∆H is positive. Oxygen Formula: O Molecular weight: 31.9988 IUPAC Standard InChI: InChI1S/O2/c1-2. For the following reaction, Delta H 572 kJ and Delta S 179 J/K. For the reaction of SO2 with O2 to form SO3 over the range. For the reaction of SO2 with O2 to form SO3 over the range of temps 300-1000K, calculate the delta H and delta S. \nonumber\]īoth ways to calculate the standard free energy change at 25 ☌ give the same numerical value (to three significant figures), and both predict that the process is nonspontaneous (not spontaneous) at room temperature (since \(ΔG^o > 0\).This looks like a homework question, so I'll give you some hints to get you on the riht path rather than answering directly.ģ) We know that ∆G = ∆H - T∆S. Answer to Solved For the reaction of SO2 with O2 to form SO3 over the.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
